
And how did we know that we have two bromides for every calcium? Well, because when calcium So it's going to be like this, Br2, and there you have it, that is the chemical formula for calcium bromide. So this is going to be, forĮvery one of the calciums, you're going to have two bromides. You only have 1- here, so you're gonna have to have two bromides for every of the calcium ions. So how is that going to happen? Well, have you 2+ here, The charge of the calcium cation is going to cancel out Has, well, we don't see any net charge here,įor an ionic compound, these things are going Is for an ionic compound, especially one that Now, what is the formula going to be, and remember, the key here It's gonna wanna gain an electron, that's what the elements Like to gain an electron to have eight electrons We see that it likes to gain an electron and so it makes sense that Sits in our periodic table, right over here, we see it is a halide. Is going to be a negative ion or it's going to be an anion. Ionizes, it is going to be, it is going to ionize as Ca2+. Losing two electrons and that's because they have two electrons in their outermost shell and Known as alkaline earth metals, they tend to ionize by Over here in Group Two, and Group Two elements, also Periodic table to confirm that it's likely that calcium So that's a pretty good clue that calcium is going All right, so theĬonvention is that we write the positive ion first and Inspired, pause the video and see if you can come Let's now see if we can come up with the chemical formula for the ionic compound calcium bromide. The elements in the middle of the periodic table, the transition metals, are odd in that they have the potential of taking on several possible positive charges. And the groups further to the right tend to take on negative charges to become anions because they like to accept electrons now instead of donate them.Ĭollectively these elements in groups 1-2 & 13-18 are known as main block elements and have (usually) constant charges. Elements in group 14 have the possibility of taking on a +4 or a -4 charge. Elements in group 13 take on a +3 charge. Elements in group 2, the alkaline earth metals, take on a +2 charge for a similar reason. 
Elements in group 1, the alkali metals, have one valence electrons so they tend to lose that one electron and take on a +1 charge. It corresponds to how many valence electrons those elements in that group have and therefore how many they wish to donate to other atoms to form cations. The table lists some additional examples.For the most part it depends on the group (column) in which the element is found in on the periodic table. The chemical formula of Magnesium Chloride is “MgCl₂”. 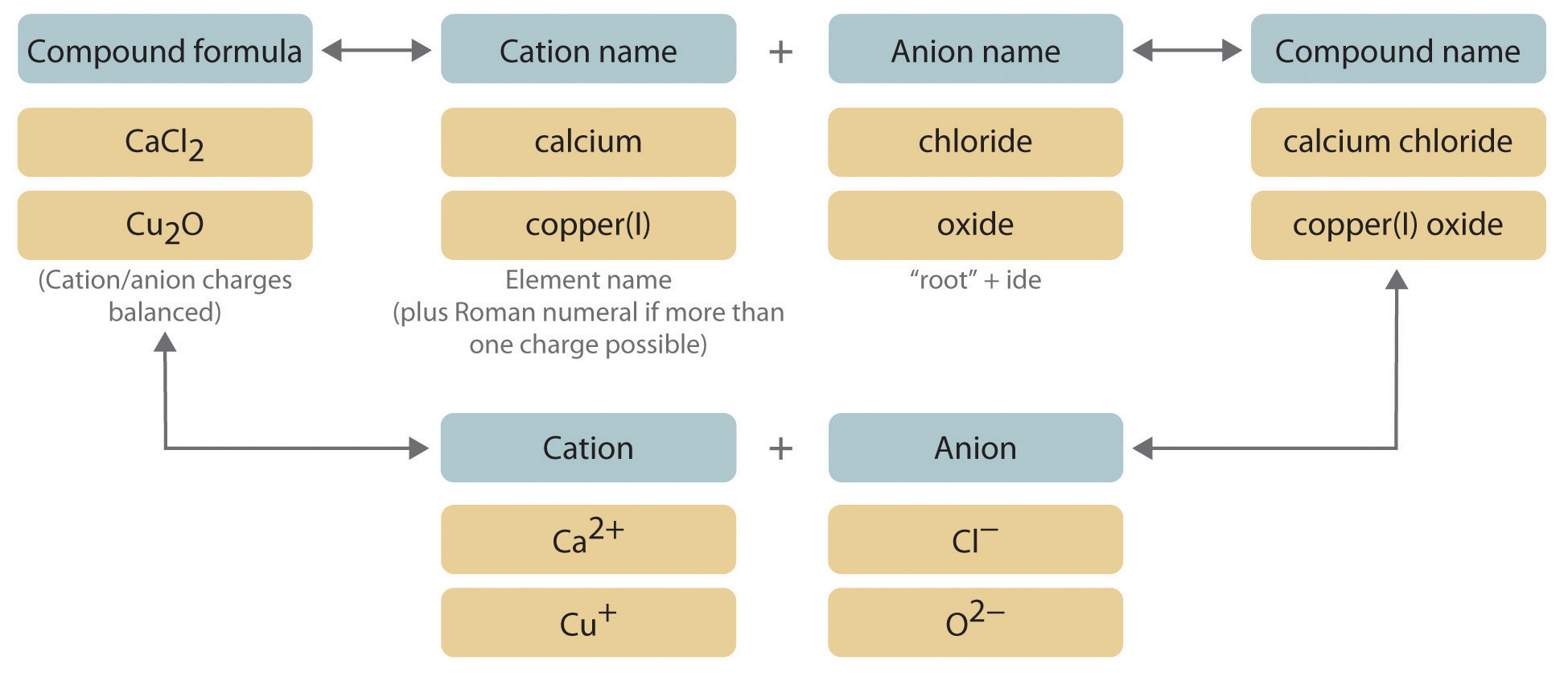
Accordingly, 2 Cl atoms can give a total negative charge of -2 in order to balance the +2 charge on Mg. Chloride refers to chlorine, whose symbol is “Cl” and has charge -1. “K” is the chemical symbol of potassium and it belongs to the 1 s t \rm 2 n d group of the periodic table. To find the chemical formula from the chemical name of the compound: When both are put together, name Calcium Oxide is obtained. “O” is the symbol for oxygen, and its root is “ox.” By adding the suffix “ide”, oxide is obtained. When both are put together, the name Rubidium Iodide is obtained. “I” is the chemical symbol for iodine, and its root is “iod.” By adding the suffix “ide”, iodide is obtained. “Rb” is the chemical symbol for rubidium. To find the chemical name from the chemical formula of the compound: Some examples will help study the naming of compounds from the chemical formula and the chemical name.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
